|
|
|
You can get e-magazine links on WhatsApp. Click here
|
|
|
|
|
|
FSSAI reveals RDA for vitamins & minerals to enable review of products
|
|
Tuesday, 28 January, 2020, 08 : 00 AM [IST]
|
|
Ashwani Maindola, New Delhi
|
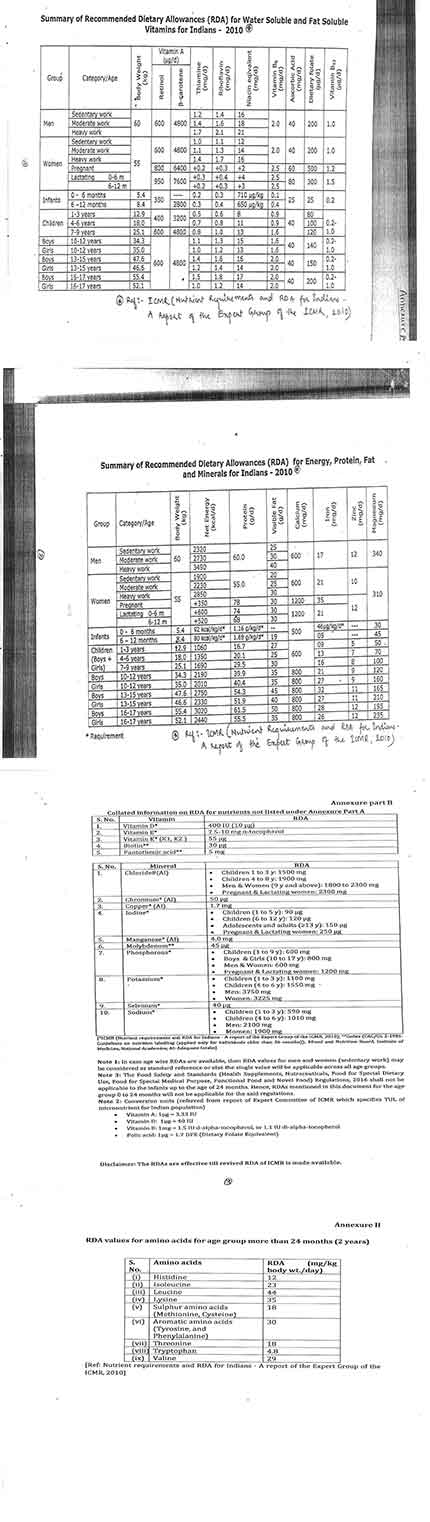
The Food Safety and Standards Authority of India (FSSAI) has prepared a document in respect of the Recommended Dietary Allowance (RDA) for vitamins and minerals so that food businesses can review their food products.
The FSSAI has also issued a separate list of essential amino acids for general population based on references from ICMR (Indian Council of Medical Research).
FSSAI says that under the Section 22 of the FSS Act, the FBOs were allowed to use vitamins, minerals, and amino acids not exceeding the RDA for Indians in functional foods, food for special dietary uses, nutraceuticals, and health supplements.
Currently the FSS-Health Supplements, Nutraceuticals, Food for Special Dietary Use, Food for Special Medical Purpose, Functional Food and Novel Food-Regulations 2016 specifies that addition of nutrients not exceeding the RDA as specified by the ICMR and in case such standards are not specified, the standards laid down by international food standards body, Codex shall apply.
FSSAI, in a statement, said that, the apex food regulator was in receipt of representation from stakeholders as well as the regulatory staff seeking clarification on RDA values of different nutrients.
“In this regard, based on the recommendation made by the scientific panel and scientific committee, a collated document in respect of RDA values for vitamins and minerals has been prepared and placed on FSSAI’s website. Further, the scientific panel has also recommended RDA values for essential amino acids for general population using references from ICMR which was endorsed by the scientific committee,” read the statement.
The apex food regulator also clarified that the tolerance upper limit of vitamins and minerals which was made available to the public through FSSAI’s website in September last year was ‘only for information and not for use’ by the FBOs in the field of health supplements and nutraceuticals.
|
|
|
|
|
|
|
|
|
|